
Melarsomine Is the First Choice Among Veterinarians for Treating Heartworm Disease in Dogs1

Getting Your Dog Back to Normal Sooner
The American Heartworm Society recommends the active ingredients in Diroban for heartworm disease treatment.1 With only 2-3 veterinarian administered doses, recovery time is far shorter than other treatments which can last
up to 2 years.
Diroban is administered by your veterinarian

Fast and Effective2
Diroban (when combined with a heartworm preventative, anti-inflammatories, and antibiotics) has been shown to effectively kill most heartworms in 1-3 months.1 Other forms of therapy can take up to 2 years, potentially causing unnecessary risks of long-term damage.3

Heartworm Disease & Your Dog
While heartworm disease is treatable, it’s better for your dog’s health to proactively protect them by using an effective preventative product.
Heartworm disease is an infestation of threadlike, parasitic worms in the heart (as well as the lungs and blood vessels) that affects dogs.
If left untreated, heartworm disease will eventually cause:
- Severe lung issues and breathing difficulty
- Damage to other organs, like the liver and kidneys
- Heart failure, in extreme cases
As a result, it is crucial to test dogs for heartworm and treat accordingly if heartworm disease is discovered. The longer heartworms live in your dog, the more damage they can do.
It’s hard to spot early signs of heartworm disease. That’s why it’s so important for your dog to be tested every single year. In the more advanced stages of the disease, symptoms include fatigue, labored breathing and coughing.4
If you see any of the following signs in your dog, contact your veterinarian immediately.

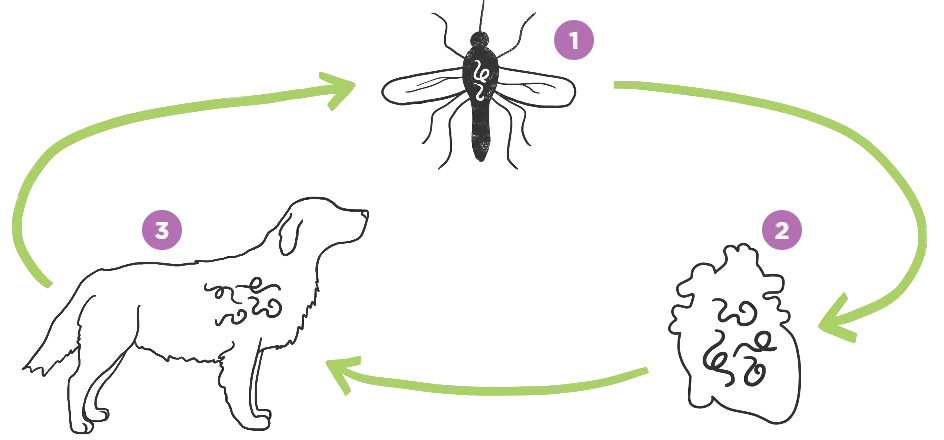
The short answer: mosquitoes. A single bite from an infected mosquito can put your dog at risk for developing heartworm disease.
- An infected mosquito bites your dog, infecting it with heartworm larvae.
- The heartworms then mature inside your dog, living in major vessels around the heart and even sometimes inside the heart itself.
- It could take longer than 7 months for the heartworms to be detectable with routine testing. An adult heartworm can grow up to a foot in length, living inside your dog’s heart and lungs.1
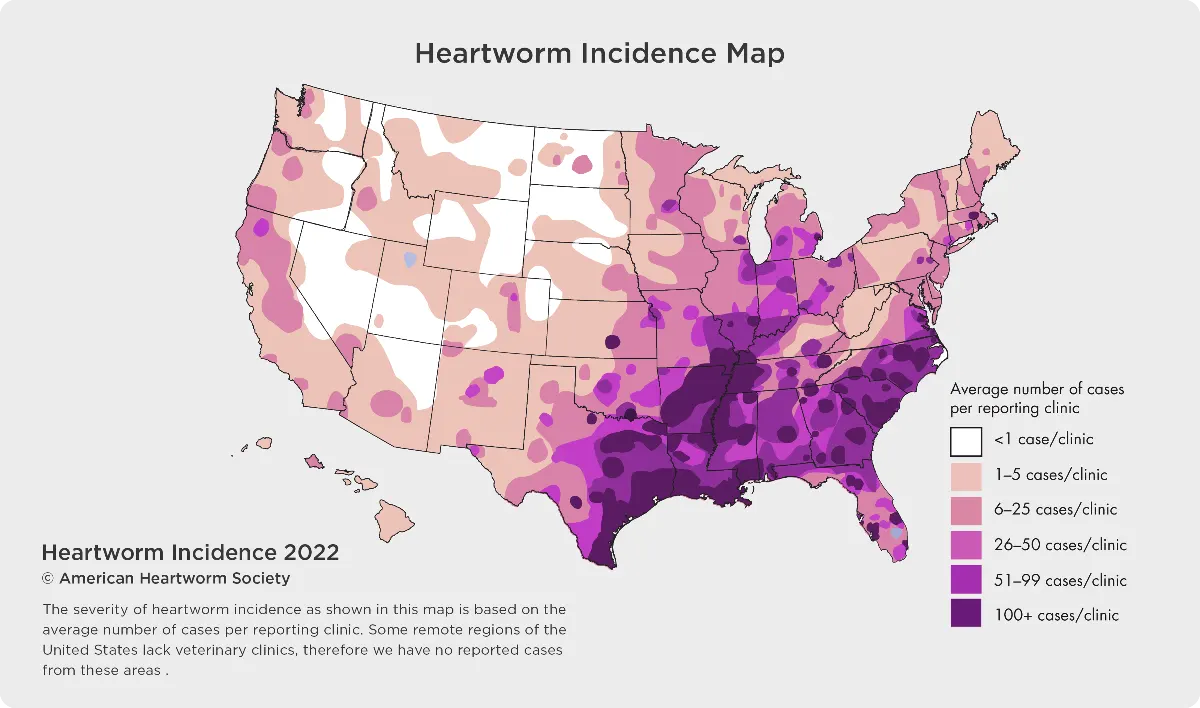
Heartworm disease has been reported in all 50 states. Climate differences and changes, as well as the presence of wildlife carriers cause infection rates to change dramatically every year. Most people don’t realize that infected mosquitoes can enter your house, so both outdoor and indoor pets are at risk.
Caring for Your Dog During and After Treatment
For up to 12 weeks after the first dose of Diroban, it will be absolutely essential to keep your dog calm. That means restricting all exercise and activity that would elevate your dog's heart rate or increase his blood pressure. That's because dead heartworms can collect around organs and cause potentially fatal blood clots.
Every case of heartworm disease is different. Your veterinarian will develop a treatment plan specifically for your dog to advise you on aftercare.

Remember that as difficult as the 12-week restricted activity period might be for your dog, its much easier than restricting your dog for up to 2 years following the slow-kill therapy method.
-
Replace Activity with Affection
Snuggle up while you’re online, reading or watching TV.
- Keep Your Dog Away From Visitors
Be conscious of both humans and animals. If they bark at outside activity, be sure to keep them away from windows.
- Give Them Plenty of Things to Chew
A bored dog is sometimes a destructive dog. Durable chew toys can channel this behavior and help keep your confined dog happily occupied.
If you are afraid that your dog will struggle under these restrictions, ask your veterinarian about options for keeping them still and calm during this critical recovery phase.
-
About 6 months after the final Diroban treatment, your veterinarian will perform a follow-up test to see if all the heartworms have been treated.
With a single mosquito bite, your dog can develop heartworm disease all over again. Give them heartworm prevention year-round to keep them protected.
Veterinarian-Approved Resources
Discover More Pest Preventatives from Zoetis
IMPORTANT SAFETY INFORMATION: See full Prescribing Information. Diroban is for use in dogs only. Do not use in dogs with very severe (Class 4) heartworm disease. Avoid human exposure. Consult a physician in cases of accidental human exposure by any route. Diroban should be administered by deep intramuscular injection in the lumbar (epaxial) muscles (L3 – L5) only. do not use in any other muscle group. do not use intravenously. Care should be taken to avoid superficial injection or leakage. Safety for use in breeding, pregnant, or lactating animals has not been determined. Common side effects include injection site irritation (accompanied by pain, swelling, tenderness and reluctance to move), coughing/gagging, depression/lethargy, anorexia/inappetence, fever, lung congestion, and vomiting. All patients should be monitored during treatment and for up to 24 hours after the last injection.
Simparica Trio
See full Prescribing Information. Use with caution in dogs with a history of seizures. Simparica Trio contains sarolaner, a member of the isoxazoline class which has been associated with neurologic adverse reactions including tremors, ataxia, and seizures in dogs with or without a history of neurologic disorders. The safe use of Simparica Trio has not been evaluated in breeding, pregnant, or lactating dogs. The most frequently reported adverse reactions in clinical trials were vomiting and diarrhea.
ProHeart 12
See full Prescribing Information. Use ProHeart 12 in dogs 12 months of age or older. Do not administer to dogs that are sick, debilitated, underweight, have a history of weight loss, or to those previously found to be hypersensitive to the drug. Hypersensitivity reactions may occur in some dogs when ProHeart 12 is administered alone or with vaccines. Anaphylactic and anaphylactoid reactions can result in death and should be treated immediately with the same measures used to treat hypersensitivity reactions to vaccines and other injectable products. The most common reported side effects in clinical trials were vomiting, lethargy, diarrhea, and anorexia. People should avoid inhalation, contact with eyes, or accidental self-injection. Certification is required before veterinarians and staff administer ProHeart 12.
References:
AHS Canine Guidelines 2024. https://d3ft8sckhnqim2.cloudfront.net/images/AHS_Canine_Guidelinesweb03FEB2025.pdf?1738626677. Accessed February 25, 2025.
DIROBAN® Sterile Powder for Injection Prescribing Information. ANADA 200-609. Zoetis.
Venco L, McCall JW, Guerrero J, Genchi C. Efficacy of long-term monthly administration of ivermectin on the progress of naturally acquired heartworm infections in dogs. Vet Parasitol. 2004;124(3-4):259-268.
Pet owner resources: Heartworm basics. American Heartworm Society. https://www.heartwormsociety.org/pet-owner-resources/heartworm-basics. Accessed February 25, 2025.


